 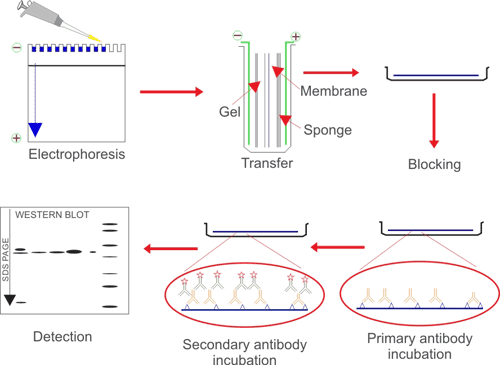
If you set it up to run overnight, it shouldn't hold you up too much. For instance, if it is reducing a disulphide bond to give a free cysteine, then you may do well to protect this cysteine or mutate it out (if this is practical or desirable).įor cleaning the column, I'd always do salt, base and acid washes, with water in between, and multiple switches between base and acid. I planed to run the samples the next day. After heating it up to 95☌ for several minutes I placed in in the fridge. Other additives might help, but it does depend on why the DTT helps. I added 4x leammli buffer to my protein samples and DTT. TCEP (tris carboxy ethyl phosphine) is another.  
To your questions, can you simply add the DTT to the sample and incubate for some time (30mins?) before applying to the column? I'd say DTT is one of the cheapest additives for generating a reducing environment. A gentle acetone wash removes TCA and co-precipitants allowing the end-user to recover the protein in a small volume of buffer. The exact composition of Protein Precipitation Solution is confidential. What pH are you using? Did you try changing the pH to try and raise the buffer strength, and have better binding? If your buffer conditions are not adequate then your proteins will be binding to your column by different mechanisms. I would say that 2mM phosphate is very close to not being a buffer at all. In elution buffer, the protein is in the supernatant and everything seems ok but when Im starting dialysis against 2M NaCl, DTT to refold protein it starts precipitating. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
